Multifunctional surface engineering of titanium alloys: tailored porosity and biocompatible coatings for controlled delivery of therapeutic agents in orthopedic applications

IBV coordinator
Uroš Maver
Duration
2025 – 2027
Back to the list
Short description
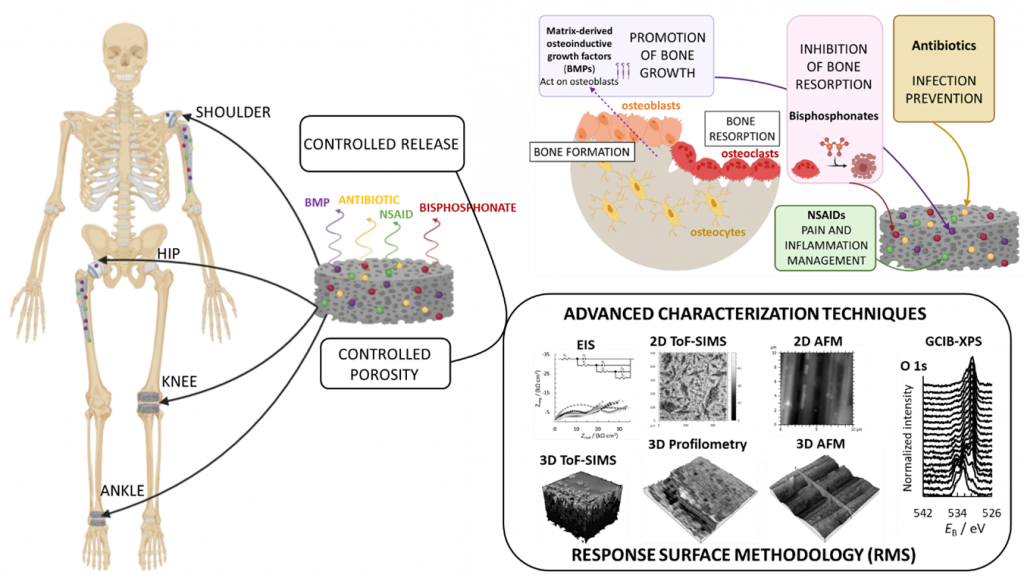
This project addresses one of the major challenges in the polymer-based delivery of therapeutic agents from metallic implant materials – the poor adhesive properties and coating stability that lead to detachment from the implant during surgical implantation. The objective is to develop a unique combination of therapeutic agents (osteoinductive growth factors, osteoclast inhibitors, antibiotics, and NSAIDs) simultaneously incorporated into a biodegradable polymeric delivery system to produce conjugates of polymer‑therapeutic agents (P-TA), enabling controlled release of patient-tailored formulations. The P‑TA conjugates will be embedded into porous 3D-printed titanium alloy substrates to prevent removal during surgical implantation. The selection of therapeutic agents will be performed with an advanced optimization procedure and combined with 3D-printing of the metal substrate, providing a personalized solution.
Metal-based implants have been used for decades and will continue to dominate the field of orthopaedics for the foreseeable future. Despite some excellent properties, these materials are not without flaws, potentially leading necessitating revision surgery during failure. A known disadvantage of metal trusses is the difference between the Young modulus of the bone and the metal (i.e., the so-called stress shielding effect). To overcome this, porous metal materials with a stiffness similar to the native bone are employed. However, the manufacturing process of the currently available porous metal materials is incapable of producing implants with controlled porosity that meet the requirements of individual bone properties. Nevertheless, such solutions based on patient-specific anatomy can be fabricated by 3D-printing and sophisticated CAD design.
The selected P-TA conjugates will enable the treatment of severe pain and inflammation (by application of NSIADs), while preventing the occurrence of “device-related” infections (by application of antibiotics). Moreover, osteoinductive growth factors and osteoclast inhibitors will provide post-operative support for improved implant fixation and prevention of implant loosening, a common cause of orthopaedic revisions, especially in elderly patients with poor bone quality. The avoidance of the latter represents the rationale for the systematic development of improved functionalized metal-based implants.
Biodegradable polymers can be used as a controllable means to deliver different therapeutic agents in a sustained manner avoiding potential unwanted side effects often associated with systemic therapy. Their use in orthopaedics has been limited due to the lack of mechanical integrity and the absence of systematic studies evaluating potential interactions between therapeutic agents, implant materials, and the biological environment. To overcome this, the P‑TA conjugates used in our study will be embedded into porous 3D-printed titanium alloy substrates with defined surface and bulk morphology, meeting the required bone repair properties. The resulting composite implant materials will be systematically investigated using a range of advanced analytical methods such as nano‑CT, ToF‑SIMS, GCIB-XPS, UHPLC-Orbitrap MS, TEM, etc. The development of modified orthopaedic implant materials is usually governed by trial-and-error experiments. After defining the problem, the proposed solution is tested, errors are analyzed, and the whole process is repeated. This approach is not only terribly time-consuming and expensive but also inefficient. Because the outcome in developing such complex materials can be affected by many factors, the experimental parameters will be optimized by Response Surface Methodology (RSM), supported by state-of-the-art characterization techniques. This will enable reverse manipulation, identification of significant experimental factors, and their optimization t